What is Callender and Barnes method?
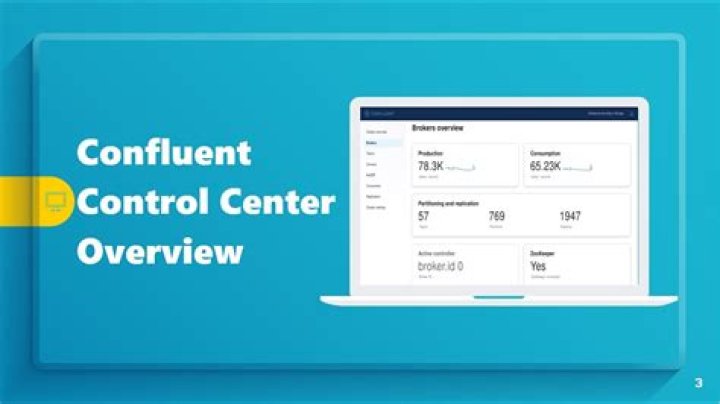
CALLENDER AND BARNE’S CONTINUOES FLOW CALORIMETER – Physics Equipment Callender and Barnes’ Apparatus also known as Continuous Flow Calorimeter is a scientific instrument that is designed to find the specific heat capacities. For determining the specific heat of water or similar liquids.
What are the advantages of Callender and Barnes apparatus?
The continuous-flow calorimeter This was first developed by Callender and Barnes in 1902 for the measurement of the specific heat capacity of a Liquid, and is shown in Figure 2. Its main advantage is that the thermal capacity of the apparatus itself need not be known.
Which of the following is the formula of calculating the mechanical heat of water?
The specific heat capacity of water is 4.18 J/g/°C. We wish to determine the value of Q – the quantity of heat. To do so, we would use the equation Q = m•C•ΔT.
What is J by electrical method?
The Joule equivalent of heat is the amount of mechanical or electrical energy contained in a unit of heat energy. The factor is to be determined in this experiment. It is an experimental observation that when a current runs through a wire, the wire will increase its temperature.
What is Joule calorimeter?
The Joules Calorimeter is for the determination of the specific heat capacity of a liquid by the electrical method. Made of nickel plated copper, the Joules Calorimeter is ideal for student experiment and demonstrations.
What is the value of specific heat of water?
The SI unit of specific heat capacity is joule per kelvin per kilogram, J⋅kg−1⋅K−1. For example, the heat required to raise the temperature of 1 kg of water by 1 K is 4184 joules, so the specific heat capacity of water is 4184 J⋅kg−1⋅K−1.
What are the 3 formulas of heat?
Different formulas for Heat
- H = (VI)t.
- H = (I 2 R)t.
- H = (V 2 /R)t.
What are the three formulas for calculating heat?
To calculate the amount of heat released in a chemical reaction, use the equation Q = mc ΔT, where Q is the heat energy transferred (in joules), m is the mass of the liquid being heated (in kilograms), c is the specific heat capacity of the liquid (joule per kilogram degrees Celsius), and ΔT is the change in …
What is Joule constant?
(Also called mechanical equivalent of heat.) The amount of mechanical work necessary to raise the temperature of a given mass of water by 1 degree Celsius. In modern usage it is the heat capacity of water, which at room temperature is 4186 Joules kg-1.
What is the CV of water?
Cv is the energy required to raise the temperature of a unit mass of a substance by one degree as the volume is held constant….Specific Heat Capacity of Liquid Water Calculator:
| KNOWN PARAMETERS | ||
|---|---|---|
| Parameter | Value | Unit |
| RESULTS | ||
| Parameter | Value | Unit |
| Specific Heat (cp) | — | J/kg · K Btu/lbm·°F kJ/kg·K kJ/kg·°C |
Why is CP is greater than CV?
The molar heat capacity at constant pressure is represented by Cp. At constant pressure, when a gas is heated, work is done to overcome the pressure and there is an expansion in the volume with an increase in the internal energy of the system. Therefore, it can be said that Cp is greater than Cv.
How do you calculate heat flow?
Heat flow is calculated using the rock thermal conductivity multiplied by the temperature gradient. The standard units are mW/m2 = milli Watts per meter squared. Thus, think of a flat plane 1 meter by 1 meter and how much energy is transferred through that plane is the amount of heat flow.
What is the function of Callendar and Barne flow?
In the Callendar and Barne flow, we have an enclosed glass tube with the heating elememt inside and 2 thermometers at the ends . The water which is constantly supplied by keeping its height in the tank contant flows through this glasstube and gets heated.
What apparatus did Callendar and Barnes use?
The apparatus that Callendar and Barnes used is shown in the Fig. 3.5. AB is a glass tube whose middle portion is very narrow. Through this narrow portion, a special platinum wire is passed which is fixed to two copper tubes of negligible resistance at its two ends.
How does a Callender-and-Barnes calorimeter work?
One of the openings is connected to the tap, the middle one to the sink, and the other to the inlet end of the Callender-and-Barnes calorimeter. The height of the reservoir is adjusted and water is allowed to flow through the tube at a constant pressure.
What is the advantage of continuous flow calorimeter?
The continuous-flow calorimeter This was first developed by Callender and Barnes in 1902 for the measurement of the specific heat capacity of a Liquid, and is shown in Figure 2. Its main advantage is that the thermal capacity of the apparatus itself need not be known.